What is the purpose of Tips-Haler Inhalation Chamber Mask for infants aged 9 months and over?
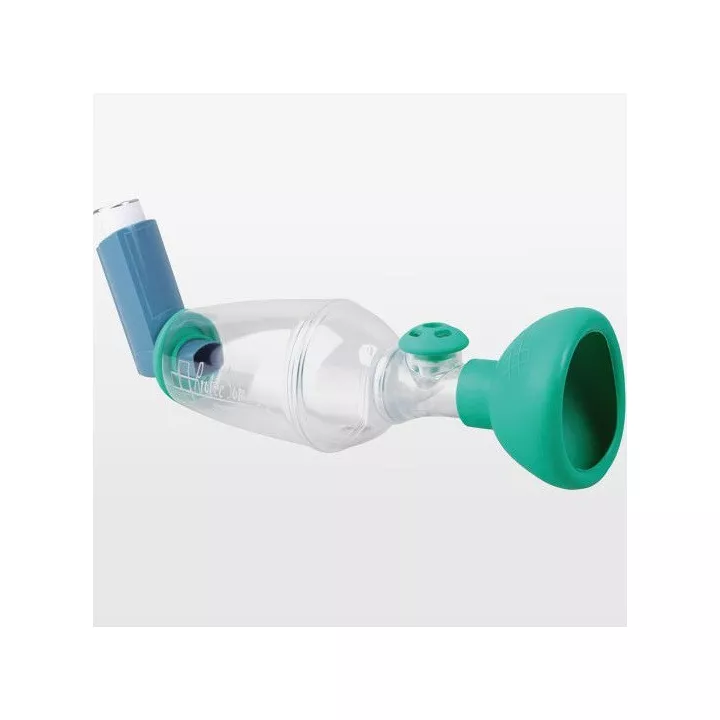
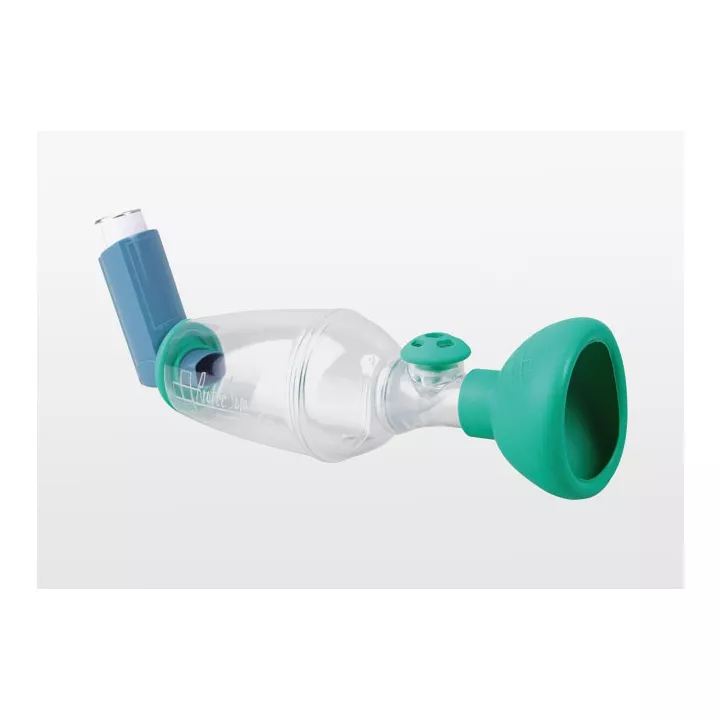
With its ISObreath® valve, Tips-Haler® enables more targeted pulmonary deposition of medication.
Designed with safe materials, Tips-Haler Chambre Inhalation Nourisson 9Mois contains no phthalates or bisphenol A, providing effective support for patients suffering from respiratory pathologies requiring the use of an Inhalation Chamber. Used for the administration of inhaled bronchodilators or corticosteroids, these inhalation chambers fit all Metered-Dose Inhalers (MDIs), ensuring accurate treatment delivery. Coarse particles are retained to prevent them impacting the throat or mouth, causing undesirable side-effects. In general, the use of inhalation chambers improves compliance and treatment efficacy in children. Thanks to its different masks adapted to the physiology of the smallest children, the Tips-haler® inhalation chamber enables optimal administration of metered-dose inhalers.
Equipped with the ISObreath® valve, the Tips-Haler® inhalation chamber guarantees more precise pulmonary deposition of inhaled medications. This innovative technology enables better targeting of the airways, ensuring optimal absorption of treatment and greater effectiveness in relieving respiratory symptoms.
Tips-Haler Chambre Inhalation Masque Nourrisson 9 Mois is made from materials that are safe for infants' health. Free from phthalates and bisphenol A, this inhalation chamber ensures safe administration of treatment, reducing the risk of adverse effects linked to exposure to harmful substances.
This inhalation chamber fits all Metered-Dose Inhaler (MDI) mouthpieces, offering great versatility of use with different types of inhaled medication. Masks specially designed for the physiology of infants enable optimal treatment delivery, ensuring maximum comfort during the inhalation session.
Use of the Tips-Haler® inhalation chamber promotes better compliance with treatment in infants, thanks to its ease of use and comfort. By enabling precise, controlled administration of the medication, this inhalation chamber guarantees optimum treatment efficacy, helping to relieve respiratory symptoms and improve the quality of life of infants suffering from respiratory pathologies.
We also recommend Uriage Bébé 1ère Trousse de Voyage, which will enable you to take care of your baby with ease, wherever you are.
How do I use this inhalation chamber?
Thanks to its optimized shape, Tips-Haler® delivers just the right dose of product, while retaining the large particles that generally impact the mouth and throat, making it the product's key asset. A clearly visible exhalation valve makes it easy to check that treatment has been taken, and deposits the medication only where it's needed.
- Compatible with all metered-dose inhaler nozzles
- Easily removable for perfect hygiene
- Ideal size for transport
What does it consist of?
Tips-haler® is a medical device guaranteed phthalate-free and Bisphenol A-free, for improved quality of use. Tips-haler® has been designed with you and your comfort in mind.
Introducing Tips-Haler Infant Inhalation Chamber
An Inhalation Chamber for Infants plus a Mask. Sold at the best price in your online pharmacy.
Tips-Haler complies with applicable French and European standards. Efficacy and safety tests are regularly carried out on manufactured batches. Traceability is ensured from batch to user, for all products manufactured. The three-year ISO 13485:2012 certification has been granted until 2014, providing patients and physicians with a guarantee of the quality of medical devices. ISO 13485:2012 is the recognized standard for the implementation of quality management system effectiveness control procedures for manufacturers and distributors of medical devices. It defines the requirements of a quality system for companies needing to demonstrate their ability to supply medical devices that comply with customer requirements and applicable regulations.